|
Since the valence electrons are in the 3rd principal quantum shell for chloride, compared to the 2nd principal quantum shell for the valence electrons of magnesium ion. We will also draw 8 electrons in the valence shell of chloride ion. Chloride ion has an electronic configuration of (2,8,8). A chloride ion has a charge of -1, and will have 18 electrons. We will draw 8 electrons in the valence shell for magnesium ion.Ī chlorine atom has 17 electrons. 
Magnesium ion has an electronic configuration of (2,8). A sodium ion which has a charge of +2 will have 10 electrons. With that, the formula of sodium chloride is MgC l 2.Ī magnesium atom has 12 electrons. Chlorine is in group VII of the periodic table, it forms ions with charge of -1. Since magnesium is in group II of the periodic table, it forms ions with charge of +2. To draw the dot-and-cross diagram of magnesium chloride, let’s first determine its formula. dot-and-cross diagram of ionic compound sodium oxide Dot-and-Cross Diagram 3 of Ionic Compound: Magnesium Chloride We will also draw 8 electrons in the valence shell of oxide ion.īoth valence electrons of oxide ion and sodium ion are in 2nd principal quantum shell, hence they should be of the same size. Oxide ion has an electronic configuration of (2,8). An oxide ion has a charge of -2, and will have 10 electrons. With that, the formula of sodium chloride is Na 2O.Īn oxygen atom has 8 electrons. Oxygen is in group VI of the periodic table, it forms ions with charge of -2. Since sodium is in group I of the periodic table, it forms ions with charge of +1. To draw the dot-and-cross diagram of sodium oxide, let’s first determine its formula. dot-and-cross diagram of ionic compound NaCl (sodium chloride) Dot-and-Cross Diagram 2 of Ionic Compound: Sodium Oxide Hence, we should draw the valence shell of chloride ion bigger than the valence shell of sodium ion. 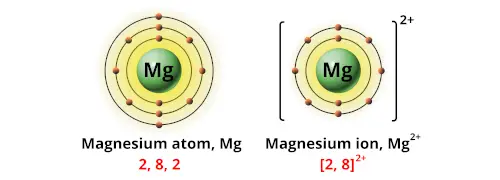
Since the valence electrons are in the 3rd principal quantum shell for chloride, compared to the 2nd principal quantum shell for the valence electrons of sodium ion. 
We will draw 8 electrons in the valence shell for sodium ion.Ī chlorine atom has 17 electrons. Sodium ion has an electronic configuration of (2,8). A sodium ion which has a charge of +1 will have 10 electrons. With that, the formula of sodium chloride is NaC l.Ī sodium atom has 11 electrons. To draw the dot-and-cross diagram of sodium chloride, let’s first determine its formula. The dot-and-cross diagrams of ionic compounds that we will be looking at will be these:ĭot-and-Cross Diagram 1 of Ionic Compound: Sodium Chloride Specifically, we will only be drawing the dot-and-cross diagrams showing the electrons in the outermost shell. In this post, let’s look at examples of dot-and-cross diagrams of ionic compounds for O Level Chemistry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
